|
|
|
| AIDScience Vol. 2, No. 4, February 2002 |
| Advances in the prevention of mother-to-child HIV-1 transmission: current issues, future challenges |
| By Marc Bulterys,1 Monica L. Nolan,1,2 Denise J. Jamieson,1 Kenneth Dominguez,1 and Mary Glenn Fowler1 |
| 1Mother-Child Transmission and Pediatric and Adolescent Studies Section, Epidemiology Branch, Division of HIV/AIDS Prevention, Centers for Disease Control and Prevention, 1600 Clifton Road, Mailstop E-45, Atlanta, Georgia 30333, United States |
| 2Projet RETRO-CI, Abidjan, Côte d’Ivoire |
| Address correspondence to: mbulterys@cdc.gov |
Abstract
| ||||||||||||||
![]() his review summarizes recent advances in the prevention of HIV-1 transmission from mother to child. In the United States and many other resource-rich countries, remarkable progress has been made since 1994 toward eliminating vertical HIV-1 transmission. Nevertheless, critical research questions as well as public health challenges remain. The U.S. Centers for Disease Control and Prevention (CDC) estimates that 280 to 370 HIV-infected infants were born in the United States each year between 1999 and 2001. New infant infections continue to occur primarily among women who did not obtain prenatal care or who were not offered HIV testing during pregnancy; innovative approaches are needed to address these barriers. Primary HIV infection in the mother during the perinatal period also contributes to infant infections. Without any intervention, the risk of mother-to-child HIV-1 transmission ranges from about 15% to 40%, with the highest rates reported in breastfeeding populations. Internationally, new clinical trial findings provide hope that a short course of treatment with antiretroviral drugs can substantially reduce vertical HIV-1 transmission in resource-poor settings in the developing world, where > 98% of all pediatric HIV infections occur. New research studies are focusing on the role of postperinatal exposure prophylaxis with antiretroviral drugs administered to the infant and/or the mother followed by early weaning and on the prevention of postnatal transmission of HIV-1 through breast milk while maintaining adequate nutrition. An urgent task now is to translate promising trial results into an effective global public health response, guided by local input and the will to maximally reduce pediatric HIV infections worldwide.
his review summarizes recent advances in the prevention of HIV-1 transmission from mother to child. In the United States and many other resource-rich countries, remarkable progress has been made since 1994 toward eliminating vertical HIV-1 transmission. Nevertheless, critical research questions as well as public health challenges remain. The U.S. Centers for Disease Control and Prevention (CDC) estimates that 280 to 370 HIV-infected infants were born in the United States each year between 1999 and 2001. New infant infections continue to occur primarily among women who did not obtain prenatal care or who were not offered HIV testing during pregnancy; innovative approaches are needed to address these barriers. Primary HIV infection in the mother during the perinatal period also contributes to infant infections. Without any intervention, the risk of mother-to-child HIV-1 transmission ranges from about 15% to 40%, with the highest rates reported in breastfeeding populations. Internationally, new clinical trial findings provide hope that a short course of treatment with antiretroviral drugs can substantially reduce vertical HIV-1 transmission in resource-poor settings in the developing world, where > 98% of all pediatric HIV infections occur. New research studies are focusing on the role of postperinatal exposure prophylaxis with antiretroviral drugs administered to the infant and/or the mother followed by early weaning and on the prevention of postnatal transmission of HIV-1 through breast milk while maintaining adequate nutrition. An urgent task now is to translate promising trial results into an effective global public health response, guided by local input and the will to maximally reduce pediatric HIV infections worldwide.
Introduction
Over the 20 years since the first cases were identified, the scale of the global HIV/AIDS epidemic has, unfortunately, far exceeded all expectations (1-3). Forty million people are currently living with HIV, and at least 22 million men, women, and children have already died.
Mother-to-child transmission (MTCT) is the dominant mode of acquisition of HIV-1 among young children worldwide, resulting in at least 1700 new infections each day or more than one pediatric infection every minute (4, 5). Globally, HIV/AIDS is one of the leading causes of death in children, and over 1 million children are living with HIV infection, primarily in sub-Saharan Africa (6). In the absence of antiretroviral (ARV) prophylaxis, the risk of transmission from an HIV-1 infected mother to her infant usually ranges from about 15% to 40%, with the highest rates reported in breastfeeding populations (7). The risk of MTCT of HIV-2, a less pathogenic HIV type common in West Africa, is only about 1% (8). Most MTCT of HIV-1 is thought to occur in late pregnancy and during labor and delivery (9, 10). Postnatal transmission through breastfeeding also causes one-third to one-half of HIV-1 infections in children worldwide (11-16). In the United States, an estimated 6000 to 7000 infants were born to HIV-infected women each year from 1989 to 1995, and more than 16,000 perinatally HIV-infected children have been born since the beginning of the epidemic (17). Over 90% of pediatric AIDS patients in the United States were infected during the perinatal period. With ARV drugs and proper obstetrical care, the risk of vertical HIV transmission has been reduced to very low levels in recent years.
There have been major advances in our understanding of the timing and pathogenesis of perinatal HIV infection in recent years, raising new hope for the prevention of perinatally acquired HIV infections (9, 10, 18, 19). The most dramatic prevention breakthrough occurred in February 1994, when the results of Pediatric AIDS Clinical Trials Group (PACTG) protocol 076 demonstrated that the risk of MTCT could be reduced from 25.5% to 8.3% by treating the mother and neonate with zidovudine (ZDV) (20).
The U.S. Public Health Service (USPHS) issued guidelines for the use of ZDV to reduce perinatal transmission in August 1994 and, in July 1995, for universal counseling and offering of voluntary HIV testing of pregnant women (21, 22). With the widespread adoption of these guidelines in the United States (23-25), the number of perinatally acquired AIDS cases has declined 81% (Figure 1). In November 2001, the USPHS issued revised guidelines for HIV screening in pregnancy emphasizing HIV testing as a routine part of prenatal care and strengthening the recommendation that all pregnant women be tested for HIV (26).
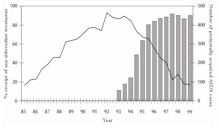 |
Figure 1. [Enlarge] Trends in perinatally acquired AIDS cases and receipt of any zidovudine (prenatal, intrapartum, neonatal). Perinatally acquired AIDS, adjusted for reporting delay and redistribution of cases reported without risk, by half year of diagnosis; receipt of any ZDV among HIV-infected women tested before or at birth in 32 HIV reporting states in the United States, 1985-1999. Data reported to the CDC through June 2000. Shaded bars: percentage of receipt of any ZDV treatment; graph line: number of perinatally acquired AIDS cases. Adapted with permission and updated from Lindegren et al. (24). |
As we enter the 21st century, it appears feasible to reduce HIV-1 MTCT rates to below 1% by treating pregnant women with potent combination ARV regimens that dramatically diminish viral load and by combining ARV treatment with elective cesarean delivery (26-31). Yet despite the effectiveness of such prevention efforts, the persistence of new cases of perinatally acquired AIDS in the United States demonstrates ongoing perinatal transmission and underscores the need for strategies to ensure that 1) women receive adequate prenatal care, timely HIV counseling, and voluntary testing; 2) HIV-infected women receive optimal ARV treatment for their own health as well as proper obstetrical care and ZDV prophylaxis to reduce perinatal transmission; 3) HIV-exposed infants receive timely postnatal ZDV prophylaxis; and 4) HIV-infected women avoid breastfeeding. Prevention efforts need to focus in particular on increasing access to care and providing prevention services to minority populations as well as women who abuse substances, among whom rates of HIV/AIDS remain the highest (32, 33).
In the developing world, simpler and less expensive ARV regimens have been identified, and rapid translation of clinical trial findings into public health policy and practice in resource-poor settings is a pressing priority (34-37). Extending the benefits of these recent breakthroughs to the majority of HIV-infected women will require innovative thinking and approaches, particularly in remote, rural communities where more than two-thirds of the population of sub-Saharan Africa resides (38). The rapid growth of periurban slum areas also presents unique challenges in extending perinatal HIV prevention. Widespread implementation of ARV interventions to prevent pediatric HIV infections in resource-poor settings provides a formidable challenge as well as a unique opportunity.
Risk and risk factors
In the absence of ARV prophylaxis, the risk of MTCT of HIV-1 appears to have decreased in a number of locations around the world. Early reports from case studies in the United States, for instance, from Miami, Florida, estimated the MTCT rate to be as high as 50% to 65% (39, 40). However, these early studies focused on HIV-infected women identified because of obvious clinical symptoms or the prior birth of a child with AIDS. Relatively high HIV-1 MTCT rates (40% to 48%) were also reported in several early cohort studies from Africa and India (41-43). In most locations where multiple prospective cohort studies have been conducted over time (e.g., France, Kenya, Malawi, Rwanda, South Africa, Thailand, and the United States), a decreasing trend appears evident, with the highest MTCT rate recorded during the early years of the HIV-1 epidemic (44-52).
Methodological and possible biological explanations for this intriguing phenomenon have been discussed in detail elsewhere (53). Whatever the exact mechanism or combination of factors responsible for an apparent decrease in HIV-1 MTCT over time, great caution should be exercised when comparing the MTCT rate in an area implementing a perinatal ARV intervention program with historical background rates.
MTCT is affected by a number of interrelated factors, not all of which have been fully elucidated. During pregnancy, the placenta provides a physical and immunologic barrier between maternal and fetal circulations and, despite HIV-1 tropism for placental cells, the transmission risk in utero is generally less than 10% and is restricted by mechanisms that have yet to be elucidated (54). Selection of maternal HIV-1 variants in the placenta probably occurs as a result of host immune selective factors (55, 56). Preliminary evidence also suggests a role for mother-child discordance in HLA-G exon 2, a major histocompatibility complex class I gene uniquely expressed in the cytotrophoblast at the maternal-fetal interface, in reduced risk of perinatal HIV-1 transmission (57).
Table 1 separates factors affecting intrauterine, peripartum, and postnatal transmission of HIV-1 into three categories based on the strength of the epidemiological evidence for each factor: 1) known risk factors for MTCT of HIV-1; 2) factors for which evidence is suggestive but not conclusive; and 3) factors for which evidence is limited or inconsistent. A detailed review of these risk factors is beyond the scope of this article; we refer the interested reader to several comprehensive reviews (40, 58-62). The relative importance of these risk factors is presently being reassessed in the context of widespread use of ARV drug regimens for perinatal prophylaxis.
Obstetric interventions to reduce transmission
Elective cesarean delivery, which is performed before the onset of labor or the rupture of membranes, has been shown to decrease perinatal transmission by more than 50% in a pooled analysis of 15 prospective cohort studies (63) as well as in a randomized clinical trial (64). Because these studies were largely conducted before the advent of combination therapy or routine use of viral load measurements, an important yet unanswered question is whether there is a benefit from elective cesarean delivery when a woman has sustained viral suppression with or without combination therapy. In a recent meta-analysis, perinatal transmission occurred in only 1% of treated women with RNA viral loads less than 1000 copies/mL (65). Given the low risk of transmission in this group, it is unclear whether cesarean delivery would provide additional benefit.
Another unanswered question is whether elective cesarean delivery can still be beneficial after labor has begun. Among women with duration of ruptured membranes < 24 hours, the risk of vertical HIV-1 transmission appears to increase approximately 2% for every additional hour in the duration of ruptured membranes (66). It remains unclear whether cesarean delivery performed after membrane rupture offers clinically significant benefit.
Washing of the lower genital tract during labor and of the newborn infant soon after delivery has been proposed as a relatively simple intervention for reducing perinatal HIV transmission. Disinfection with an agent that inactivates HIV in order to decrease neonatal exposure to infected secretions or blood may be an appropriate intervention in settings without access to elective cesarean delivery. Unfortunately, two trials using intrapartum washing with chlorhexidine have failed to demonstrate an overall effect on perinatal HIV transmission (46, 67). Although there was no overall effect, in each of the two studies a benefit was found among a particular subgroup of women. In the study by Biggar et al. (46), a beneficial effect was apparent among women who were ruptured for more than 4 hours, and in the study by Gaillard et al. (67), a benefit was found among women who douched with 0.4% chlorhexidine solely before rupture of the membranes. Another possible agent for vaginal cleansing, benzalkonium chloride, has been evaluated in a small trial in West Africa and found to be safe with few side effects (68).
Chorioamnionitis has been associated with an increased risk of MTCT in some studies (69, 71) but not in others (72, 73). It has been proposed that treatment of chorioamnionitis with antibiotics may help reduce the risk of perinatal HIV-1 transmission (74). A trial of prepartum and intrapartum antibiotic prophylaxis of chorioamnionitis with metronidazole and erythromycin is going on in Malawi and Zambia.
Numerous procedures are performed during labor and delivery that might be expected to increase fetal exposure to maternal blood and secretions and/or violate the integrity of neonatal skin or mucosal barriers. Examples include invasive procedures such as fetal scalp sampling, amniocentesis, fetal scalp electrode placement, episiotomy, and instrumental delivery by forceps or vacuum. Several studies have not found an increased risk associated with invasive intrapartum procedures (75-78). Although the French Pediatric HIV Study Group (79, 80) found that bloody amniotic fluid at delivery was associated with increased risk of perinatal transmission, specific events during labor and delivery such as fetal skin lesions associated with scalp electrode placement or scalp sampling, instrumental delivery, episiotomy or perineal lacerations, and breech extraction were not. Another study found that events associated with maternal blood exposure during labor, such as abruption or fetal scalp electrode placement, were significantly associated with perinatal transmission (81). In this study, episiotomy or perineal laceration was shown to be a risk factor for perinatal HIV transmission, but this association did not reach statistical significance. In general, the relative infrequency of many obstetric procedures may have limited the ability of researchers to adequately assess their risk for MTCT.
Perinatal antiretroviral therapy interventions
The primary focus of strategies to prevent MTCT of HIV has been on the use of perinatal ARV drug prophylaxis. Table 2 lists completed phase II/III randomized clinical trials, the ARV drug regimens utilized, and their relative efficacy in reducing perinatal HIV transmission.
In PACTG protocol 076, therapy consisted of oral administration of ZDV to HIV-infected, nonbreastfeeding, pregnant women starting at 14 to 34 weeks gestation, intravenous administration of ZDV to the mother during labor and delivery, and 6 weeks of postnatal treatment of the infant with oral ZDV (20). Although the risk of vertical HIV transmission was directly correlated with maternal viral load, the treatment with ZDV produced a median reduction of only 0.24 log in plasma HIV RNA levels; however, ZDV was effective regardless of the HIV RNA level or the CD4+ count at trial entry (82). The only observed short-term toxicity in ZDV-exposed infants was anemia (83). No adverse effects were observed in HIV-uninfected children with in utero and neonatal exposure to ZDV who were followed for as long as 5.6 years (84). In PACTG protocol 185, the 076 regimen along with either hyperimmune HIV immunoglobulin or regular immunoglobulin was administered to women with more advanced HIV disease, resulting in a perinatal transmission rate of only 5% (85).
In Bangkok, Thailand, a short course of twice-daily ZDV given orally beginning at 36 weeks of gestation and during labor to nonbreastfeeding women lowered the risk of MTCT by half compared with a placebo (50, 86). A slightly lower treatment efficacy was observed in breastfeeding populations in two clinical trials in West Africa (87, 88). In a pooled analysis of these two trials, treatment efficacy appeared to occur entirely among the group of women with relatively high CD4 counts in pregnancy (89). In a four-arm trial of ZDV regimens in Thailand, researchers found that the rate of in utero transmission was significantly higher with the two regimens when maternal treatment started at 35 weeks’ gestation (5.1%) than with the two with longer maternal treatment (1.6%) (90).
Findings from the PETRA trial, a placebo-controlled study of ZDV and lamivudine (3TC) among women in South Africa, Tanzania, and Uganda, also showed a substantial reduction in perinatal HIV transmission when ARV prophylaxis was given at 36 weeks, during labor, and for 1 week postpartum to the mother and child as well as when given intrapartum and for 1 week postpartum (91, 92). When given intrapartum only, ZDV/3TC had no effect on MTCT (Table 2).
In a clinical trial in Uganda (HIVNET 012), a single-dose nevirapine (NVP)-only regimen to mother and infant as prophylaxis against intrapartum and early transmission via breast milk reduced the transmission rate to 13% by 14 weeks of age in the NVP group, compared with 25% in the group receiving intrapartum and 1 week of neonatal ZDV (p < 0.001) (93). It should be noted that in HIVNET 012, the first dose of NVP was given to women to take home with them at about 36 weeks gestation and was self-administered at onset of labor pains, prior to admission to the hospital. NVP-resistant HIV-1 was detected at 6 weeks postpartum in a substantial number of treated women and infants in this study, but it faded from detection within 12 to 24 months (94).
The SAINT trial in South Africa confirmed the efficacy of both the HIVNET and PETRA trial arm B regimen (95). Long-term follow-up data from a pooled analysis of the West African studies (96) and the HIVNET study (97) indicate that the benefit of the different ARV regimens is maintained in infants exposed to HIV through breastfeeding up to 24 months of age.
Safety concerns with ARV therapy
Long-term safety for children exposed to ZDV, NVP, or combination ARV drugs perinatally has not yet been determined. In rodent studies, noninvasive vaginal squamous epithelial tumors and an increase in the incidence of liver and lung tumors have been described in the offspring of animals following high-dose perinatal administration of ZDV (98, 99). The relevance of rodent data to humans is unknown. In an analysis of two large U.S. cohorts, no tumors of any nature were observed in 727 children with known ZDV exposure who were followed for more than 3 years (100). Current USPHS recommendations for the use of ARV drugs among HIV-infected pregnant women in the United States state that pregnancy should not be a reason to defer standard combination ARV therapy if indicated for the health of the woman (27). Although the known benefits of highly active antiretroviral therapy for treatment of maternal HIV disease appear to outweigh risks observed to date, continued active surveillance of short- and long-term adverse effects will be imperative.
In February 1999, French researchers reported two cases of progressive neurologic disease and death among uninfected infants exposed to perinatal ZDV/3TC. Laboratory studies of these children suggested mitochondrial dysfunction (101). Subsequently, the investigators reported on six other living, uninfected children exposed to either perinatal ZDV alone or ZDV/3TC who showed clinical or laboratory findings suggestive of mitochondrial dysfunction (102). To date no similar findings of severe neurologic problems leading to death have been reported in a careful review of all deaths among HIV-uninfected children followed in U.S. perinatal HIV cohorts (103). A review of living children in the Perinatal AIDS Collaborative Transmission Study (PACTS) also found no cases of HIV-uninfected children, perinatally exposed to nucleoside analogs, with symptoms attributable to mitochondrial dysfunction (104). Nevertheless, the French cases underscore the importance of close, long-term monitoring of children exposed to perinatal ARV therapy.
Prevention of postnatal HIV-1 transmission
Innovative approaches to diminish postnatal transmission through breast milk are urgently needed. In Côte d’Ivoire, Ekpini et al. (105) found that 12% of breastfed children of HIV-1-positive mothers, who escaped HIV infection during the first 6 months of life, became HIV infected by 2 years of age. An international pooled analysis yielded an overall incidence of late postnatal transmission, defined as infection occurring after 2.5 months of age, of 3.2 per 100 child-years of breastfeeding (106). In the Nairobi randomized trial, the frequency of HIV-1 transmission via breast milk was 16%, and the majority of postnatal infections occurred in the first 6 months of breastfeeding (51, 107). Mortality rates among women randomized to the breastfeeding arm were also increased (108). Observational data from South Africa indicate that mixed feeding may be associated with a higher risk of HIV-1 transmission than either exclusive breastfeeding or exclusive formula feeding (109, 110).
Clinical risk factors include duration of breast feeding, HIV-1 seroconversion during the breastfeeding period, (subclinical) mastitis, cracked or bleeding nipples, breast abscess, young maternal age, low parity, and oral candidiasis/thrush in the infant (11, 15, 111, 112). The most important laboratory risk factors related to transmission via breast milk appear to be detectable virus and viral load in breast milk (113, 114). The possible role of immune factors in modulating breast milk infectivity is less well understood. Neonatal T-helper cell responses to HIV envelope peptides appear protective against early HIV transmission via breastfeeding (115, 116).
A number of strategies have been proposed to reduce the risk of transmission via breastfeeding for those women who choose to breastfeed. Exclusive breastfeeding for 4 to 6 months followed by abrupt weaning is being promoted in some African settings as a practical and low-cost approach to reduce postnatal HIV transmission. Trials are planned or under way that will assess whether infant or maternal ARV prophylaxis followed by early weaning, active or passive immunotherapy, or combined strategies may substantially reduce the risk of postnatal transmission. Findings from these studies should help inform optimal infant feeding choices in resource-limited settings (117). We refer the interested reader to a comprehensive AIDScience review of problems and advances in reducing transmission of HIV through breastfeeding in the developing world (118).
Behavioral interventions to reduce transmission
In two African cohort studies, investigators noted a significantly higher risk of MTCT related to heterosexual behavior around the time of pregnancy. In Butare, Rwanda, unprotected sexual intercourse with multiple partners before and during pregnancy was strongly associated with MTCT, even after adjustment for maternal immunological status and evidence of genital infection during pregnancy (119). In Brazzaville, Congo, women who had only a brief relationship with the infant’s father were at substantially higher risk of transmitting HIV to their infant, independent of the presence of HIV-related symptoms in the mother (120). In two U.S. cohorts (121, 122), a high frequency of unprotected sexual intercourse during pregnancy was associated with an increased risk of perinatal transmission even after adjustment for multiple potential confounding variables. A dose-response effect was present in the New York City Collaborative Study (121). Burns et al. (123) also showed that the association between frequency of vaginal intercourse and vertical HIV transmission appeared to be independent of maternal viral load.
Although the exact mechanism to explain these findings remains unknown, three general pathophysiologic mechanisms appear plausible. First, unprotected intercourse might increase the concentration or strain diversity (i.e., fetotropic strain) of HIV-1, particularly in the birth canal, where ejaculated virus could be partially sequestered (124-126). Viral recombination or transcriptional effects of HIV-1 superinfection could be involved (127-130). Second, frequent intercourse might increase inflammation of the cervix or vagina, either by microabrasions or, if unprotected, by sexually transmitted infections (131). Third, frequent intercourse might increase the risk of chorioamnionitis or otherwise alter the integrity of the placenta. Whatever the mechanism by which unprotected sexual activity during pregnancy increases the risk of MTCT, even if only as a surrogate for the true pathophysiology, these findings can reasonably be used to promote safer sexual practices—including condom use and avoidance of new sexual partners during pregnancy—to further reduce perinatal HIV-1 transmission, especially among populations with high HIV and STD incidence rates (132).
Perinatal HIV elimination efforts in the United States
In 1999, the U.S. Congress provided CDC with $10 million to fund domestic perinatal HIV elimination efforts. These funds were distributed to state health departments in high-prevalence states for both programmatic efforts and enhanced perinatal HIV surveillance activities and to national health care provider organizations (133).
The programmatic component involves 16 states with the highest HIV-1 seroprevalence among women of childbearing age. These funds are being used to improve linkages between agencies and organizations interfacing with women of childbearing age at high risk for HIV infection. Each state is focusing on activities that help fill the gaps in the "cascade" of services that are required to reduce perinatal HIV transmission (Figure 2). The activities fit into five main categories: 1) rapid HIV testing and other services for women with little or no prenatal care; 2) provider training; 3) outreach to high-risk women; 4) case management for high-risk women; and 5) social marketing activities. States are encouraged to collaborate so that they can build on strategies that have been effective in other states. In addition, some states are developing policies that will enhance the standard of care for HIV-infected pregnant women as well as routine HIV counseling and testing efforts.
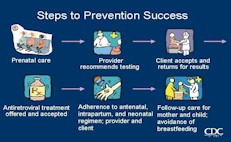 |
Figure 2. [Enlarge] Cascade of success in a perinatal HIV prevention program, United States. Adapted with permission (168). |
The enhanced perinatal HIV surveillance activities involve 21 states and four cities and are especially important for those states where HIV reporting is not mandated by law. Population-based data are being collected to better target and evaluate perinatal HIV prevention efforts locally.
The national organizations component includes the American Academy of Pediatrics, the American College of Obstetricians and Gynecologists, the Association of Maternal and Child Health Programs, CityMatCH, the National Pediatric & Family HIV Research Center, and the AIDS Alliance for Children, Youth, and Families. Most of their activities focus on improving provider training, developing educational materials for pregnant women and health care providers, conducting learning clusters and building capacity among local health departments and Ryan White Title IV grantees, and evaluating programs for rapid HIV testing in the labor and delivery setting.
Despite widespread implementation of perinatal ARV drug prophylaxis in the United States, rates of perinatal HIV transmission remain unacceptably high. Inadequate receipt of prenatal care now appears to be the major impediment to achieving lower rates (25, 134, 135). Primary HIV infection in the mother also contributes a sizable proportion of pediatric HIV cases. Epidemiological evidence from New York (136-138) indicates a likely benefit of ZDV prophylaxis postperinatal exposure, if started within 12 to 24 hours after birth, in preventing peripartum HIV infection, even in cases where prior opportunities for intervention had been missed. Support for the efficacy of infant postexposure prophylaxis also comes from experimental studies in macaques (139-141).
If pediatric HIV infection is to be maximally reduced, innovative approaches must be developed that address the needs of mothers who receive little or no prenatal care or are not offered HIV testing prenatally. Most, if not all, HIV-infected pregnant women in the United States utilize hospitals for delivery, providing a crucial opportunity for rapid screening and intervention (142). It is therefore imperative to determine whether vertical HIV transmission can be further reduced by identifying HIV-positive women entering labor with unknown status through the use of rapid testing technology (143), and providing subsequent ARV chemoprophylaxis to those who test positive and their neonates.
The Mother Infant Rapid Intervention At Delivery (MIRIAD) study is a recently initiated multicenter project, funded through CDC, focusing on disadvantaged communities in which many women receive inadequate prenatal care and exhibit relatively high HIV-1 seroprevalence (144). Funded sites are located in five U.S. metropolitan areas: Atlanta, Chicago, Miami, New Orleans, and New York City. The MIRIAD study aims to evaluate 1) innovative approaches for a 24-hour counseling and voluntary rapid HIV testing program among women in labor presenting with unknown HIV status; 2) the feasibility of obtaining informed consent during labor (or, if not feasible, soon after birth); 3) reasons for lack of prenatal care and/or HIV testing among these women; 4) the rapid implementation and assessment of ARV therapy given at labor and delivery and/or to the neonate; 5) adherence to neonatal therapy; and 6) subsequent receipt of ARV treatment and other services for women identified as HIV infected. Novel approaches for rapid bedside testing of women in labor and posttest counseling and referral to care for HIV-positive women and their infants are piloted at each of the participating hospitals.
In cost-effectiveness analyses, rapid HIV testing at labor and delivery appears to save the medical system money when HIV prevalence exceeds 0.7% to 1.0% and treatment efficacy exceeds a 5% reduction in perinatal transmission (145, 146). The MIRIAD study will evaluate the ability to obtain test samples and do rapid testing in the laboratory and/or at the bedside, the rapidity with which results are available, the efficiency of ordering and administering ARV medication, and the ability to have a sufficient duration of treatment to be effective. The MIRIAD study will also employ a behavioral postpartum intervention designed to support recently identified HIV-infected mothers to focus their attention on following HIV therapeutic guidelines for themselves and their exposed children. It is hoped that results from these studies will lead to best practice recommendations on rapid HIV testing and administration of ARV drugs among late-registrant women.
The goal of maximally reducing the number of new perinatal HIV infections in the United States appears within reach (26, 135). Remaining challenges include encouraging providers to routinely offer HIV testing to all pregnant women, improving outreach and increasing the numbers of high-risk women who access prenatal care, increasing adherence and drug treatment for substance-using HIV-positive women, instituting routine HIV retesting in the third trimester to detect primary HIV infections, and assessing the feasibility of offering rapid testing at labor/delivery to those women who have not received any prenatal testing.
Perinatal HIV prevention efforts in the developing world
In trials conducted in sub-Saharan Africa among predominantly breastfeeding HIV-1 infected women, various short-course regimens of AZT, AZT/3TC, and NVP resulted in substantial reductions in perinatal HIV-1 transmission (Table 2). Because these results represent relatively simple and inexpensive interventions that could be implemented in settings with minimal health care infrastructure, there have been substantial efforts to translate these research findings into large-scale programs around the world (34, 147). However, although the research trials focused on the narrow objective of prevention of MTCT from already infected women to their infants, it has been recognized that these implementation efforts could also contribute to other important public health goals (35, 148). Objectives of MTCT prevention programs also include promoting maternal and child health, preventing primary HIV infection among women, and assisting HIV-infected women to disclose their HIV status to their sexual partners and linking them to family planning and other medical care and social services.
Twenty years into the epidemic, millions of young people still know little, if anything, about HIV/AIDS (5). The prevention of primary HIV infection among young women in the early years of sexual activity (149-152) directly contributes to the overarching goal to reduce pediatric HIV-related morbidity and mortality.
Progress has been made toward implementing pilot MTCT prevention programs in a number of developing countries. New collaborations have been established between national governments, United Nations agencies, the international perinatal research community, major public and private donors and foundations, the pharmaceutical industry, and nongovernmental, religious, and community-based organizations. Technical and financial resources have been targeted to initiate pilot programs and then to build on these pilots to expand these programs to achieve wider population coverage. For instance, in Southeast Asia, Thailand has provided an exemplary model of learning from large-scale pilot programs and rapidly expanding to the national scale (153). Botswana, Brazil, and Uganda are providing relatively successful models of large-scale MTCT prevention programs and thus are beacons of hope for other countries. Nevertheless, lack of adequate commitment to HIV prevention by national governments remains a major impediment in many of the worst affected areas (154, 155).
MTCT prevention programs are being implemented in settings with enormous geographic, cultural, and socioeconomic diversity, and although these provide context-specific challenges, there are many common characteristics of these programs. Table 3 summarizes the essential components required to establish an MTCT prevention program.
Through these initial program implementation activities, it has been possible to identify ways to strengthen programs in the areas of counseling, rapid HIV testing, community involvement, and providing complementary services for HIV-infected women and their families. Providing high-quality HIV counseling represents a major challenge for staff and resources in most MTCT prevention pilot programs. The quality of staff-client interactions clearly affects client participation, and this needs to be addressed through provider training for effective prevention of MTCT (156). Intensive individual pre-test counseling is not possible in many settings, and quality alternatives can be successfully implemented, including the use of group pretest counseling, educational videos, and peer counselors. Figure 3 shows that in Abidjan, Côte d’Ivoire, providing rapid HIV testing and same-day test results dramatically improved posttest counseling uptake and the proportion of women who received their test results. Involvement of male partners, community leaders, and the community is increasingly recognized as an essential MTCT prevention program component (157). Strategies that target these groups have the potential to improve general knowledge about HIV and MTCT interventions, to improve HIV-test acceptance of women and their partners and the ability of women to safely disclose their HIV status to others, and to decrease HIV-related stigma and discrimination.
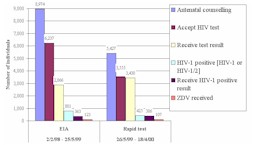 |
Figure 3. [Enlarge] Case study of change in uptake with rapid HIV test introduction, Koumassi Clinic, Abidjan, Côte d'Ivoire: standard EIA testing (2 February 1998 - 25 May 1999) versus rapid test with same-day results (26 May 1999 - 18 April 2000). Unpublished data from Projet Retro-CI (with permission from Nolan and Ekpini) |
The identification of HIV-infected women also requires improved links to a range of comprehensive health and social services. Women at highest risk for HIV-related morbidity and mortality (i.e., those with low CD4 counts) may need combination ARV treatment in order to reduce MTCT substantially, because short-course ZDV does not appear as effective in reducing transmission in this subgroup (89). In addition, the need to consider providing combination ARV treatment (158)—for her own health and survival and not just to decrease MTCT—is part of a picture that is now coming into focus. As Rosenfield et al. (159) put it, "should we not value saving women’s lives as an equal priority to decreasing transmission to infants?" The new "MTCT-Plus" Initiative is designed to support the provision of HIV-specific care, including access to standardized ARV therapy options when clinically indicated, to HIV-infected women and their HIV-infected family members (160).
Thailand provides a highly successful model of MTCT implementation. In Region 7 in Northeast Thailand, counseling was provided during a 2-year pilot period to 122,000 pregnant women, and there was a high HIV-test acceptance rate (86%); among those testing positive, 69% received ZDV (153). In sub-Saharan Africa during 1999-2000, United Nations-sponsored pilot programs were conducted on a smaller scale in nine countries with high HIV-1 prevalence; 82,000 women received prenatal care across all sites, and HIV prevalence among those tested varied from 13% to 45% with a mean of 19% (161). Of all the women receiving prenatal care, 43% accepted an HIV test (Figure 4). Of the nearly 7000 women who tested HIV-positive, 39% were documented to have received ARV prophylaxis. India has also implemented a pilot program among 125,000 pregnant women and has recorded testing and ARV prophylaxis acceptance rates comparable to the African rates. Since early 2000, several private foundations, such as the Elizabeth Glaser Pediatric AIDS Foundation and the Doris Duke Foundation, have also funded pilot perinatal HIV prevention programs in a number of developing countries.
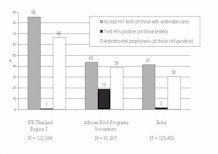 |
Figure 4. [Enlarge] Summary of implementation data from pilot programs to prevent HIV-1 MTCT, supported by the United Nations, 1998-2000: Region 7 Thailand, pilot programs in nine sub-Saharan African countries, and India. |
Experience from these pilot programs highlights three important implementation challenges: 1) MTCT intervention programs need to be well integrated within existing maternal and child health services. 2) In many countries, less than 1% of HIV-infected women currently have access to interventions to prevent MTCT; therefore, the need is great to scale up pilot programs quickly to make these services accessible to the large populations at risk. 3) There is an urgent need to raise social support and community awareness and improve overall HIV testing acceptance and other services among HIV-infected women and their partners; top-level political commitment is paramount. Some pilot programs in diverse settings in Asia, Latin America, Eastern Europe, and sub-Saharan Africa have achieved similarly high testing and ARV drug acceptance results to those documented in Thailand, so it appears that there are no insurmountable cultural or socioeconomic obstacles to high rates of testing and ARV drug coverage.
However, other challenges for international MTCT prevention efforts are more difficult to address, including issues around infant feeding, partner involvement, and the provision of care to HIV-infected persons. The reduction of postnatal HIV transmission through breast milk is particularly problematic in many resource-poor settings, as there are limited data on which to base recommendations and there are competing risks which are individual and context specific (117). In resource-poor settings with high infant and maternal mortality, breastfeeding protects against substantial health risks. Where breastfeeding is common and HIV is highly stigmatized, not breastfeeding can be perceived by others to be indicative of HIV infection and places the woman at risk of social harms due to discrimination. Furthermore, the experience of decades of work in breastfeeding promotion has shown that any shift in breastfeeding practices is slow and hard won (162). In many resource-poor areas, there are few health care providers with the time, knowledge, and skills to provide infant feeding counseling, support optimal breastfeeding practices and early weaning, and promote appropriate nutritional supplementation or replacement feeds.
Collaborations and partnerships continue to evolve. Increasingly, prevention of perinatal HIV transmission is on the national and international political agenda (163, 164). The perinatal research community has assisted the initial development of training and program guidelines. The contributions of private foundations, the United Nations, bilateral organizations, and the pharmaceutical industry have reduced some of the financial barriers. In 1999, CDC established the Global AIDS Program (GAP) to implement international HIV prevention, care, and infrastructure support, in collaboration with other federal agencies, national ministries of health, and U.N. agencies (3). GAP currently provides support in 24 countries and emphasizes sustaining intervention programs for primary prevention of HIV infection (including MTCT), developing infrastructure and laboratory support, and providing home- and community-based care for persons living with HIV.
Conclusion
Two patterns of HIV-1 MTCT now exist (34, 165, 166). In industrialized nations, dramatic declines in perinatal HIV infections were observed after integration of prenatal HIV counseling and testing into prenatal care and widespread implementation of ARV prophylaxis. Reducing the MTCT rate to below 1% appears within reach. By contrast, many resource-poor countries face a perinatal HIV epidemic as a result of increasingly devastating levels of HIV infection, a crumbling maternal and child health care infrastructure, and limited HIV counseling and testing programs. Clinical trials and observational epidemiologic studies conducted in the developing and the developed world are leading to the development of practical, affordable, and innovative approaches to reduce infant HIV infections worldwide, including in populations where prolonged breastfeeding is currently the norm. The results of these research efforts must lead to coordinated public health action and a global commitment to extend the benefits of perinatal HIV prevention to families living in resource-limited settings.
References
| 1. | P. Piot, M. Bartos, P. D. Ghys, N. Walker, B. Schwartlander, Nature 410, 968 (2001). |
| 2. | K. A. Sepkowitz, N. Engl. J. Med. 344, 1764 (2001). |
| 3. | "The global HIV and AIDS epidemic," MMWR 50, 434 (2001). Available online. |
| 4. | "HIV in Pregnancy: A Review," World Health Organization and United Nations Programme on AIDS [online], July 1999. Available online. |
| 5. | "AIDS Epidemic Update," United Nations Programme on AIDS (UNAIDS) [online], December, 2001. Available online. |
| 6. | A. Nicoll, I. Timaeus, R. M. Kigadye, G. Walraven, J. Killewo, AIDS 8, 995 (1994). |
| 7. | Working Group on Mother-to-Child Transmission of HIV, J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 8, 506 (1995). |
| 8. | G. Adjorlolo-Johnson, et al., JAMA 272, 462 (1994). |
| 9. | M. Newell, AIDS 12, 831 (1998). |
| 10. | A. P. Kourtis, M. Bulterys, S. R. Nesheim, F. K. Lee, JAMA 285, 709 (2001). |
| 11. | P. Van de Perre, et al., N. Engl. J. Med. 325, 593 (1991). |
| 12. | J. Bertolli, et al., J. Infect. Dis. 174, 722 (1996). |
| 13. | J. Kreiss, Acta Paediatr. 421(Suppl), 113 (1997). |
| 14. | L. Kuhn, Z. Stein, Am. J. Public Health 87, 927 (1997). |
| 15. | J. E. Embree, et al., AIDS 14, 2535 (2000). |
| 16. | G. C. John, B. A. Richardson, R. W. Nduati, D. Mbori-Ngacha, J. K. Kreiss, East Afr. Med. J. 78, 75 (2001). |
| 17. | R. H. Byers, M. B. Caldwell, S. Davis, M. Gwinn, M. L. Lindegren, Stat. Med. 17, 169 (1998). |
| 18. | D. T. Dunn, et al., AIDS 14, 1421 (2000). |
| 19. | L. M. Mofenson, Pediatrics 106, E88 (2000). Available online. |
| 20. | E. M. Connor, et al., N. Engl. J. Med. 331, 1173 (1994). |
| 21. | "Recommendations of the Public Health Service Task Force on use of zidovudine to reduce perinatal transmission of human immunodeficiency virus," MMWR 43(RR11), 1 (1994). Available online. |
| 22. | "U.S. Public Health Service recommendations for human immunodeficiency virus counseling and voluntary testing for pregnant women," MMWR 44(RR07), 1 (1995). Available online. |
| 23. | M. L. Lindegren, et al., JAMA 282, 531 (1999). |
| 24. | M. L. Lindegren, S. Steinberg, R. H. Byers, Pediatr. Clin. North. Am. 47, 1 (2000). |
| 25. | P. M. Wortley, M. L. Lindegren, P. L. Fleming, MMWR 50(RR06), 17 (2001). Available online. |
| 26. | "Revised recommendations for HIV screening of pregnant women," MMWR 50(RR19), 59 (2001). Available online. |
| 27. | "Public Health Service Task Force recommendations for the use of antiretroviral drugs in pregnant women infected with HIV-1 for maternal health and for reducing perinatal HIV-1 transmission in the United States," MMWR 47(RR-2), 1 (1998). Available online. |
| 28. | E. R. Cooper, M. Charurat, D. N. Burns, W. Blattner, R. Hoff, J. Acquir. Immune Defic. Syndr. 24, 45 (2001). |
| 29. | A. Dorenbaum, abstract LB7A presented at the 8th Conference on Retrovirus and Opportunistic Infections, Chicago, February 2001. |
| 30. | European Collaborative Study, AIDS 15, 761 (2001). |
| 31. | L. Mandelbrot, et al., JAMA 285, 2083 (2001). |
| 32. | M. Bulterys, M. G. Fowler, Pediatr. Clin. North. Am. 47, 241 (2000). |
| 33. | S. L. Orloff, et al., J. Acquir. Immune Defic. Syndr. 28, 65 (2001). |
| 34. | K. M. DeCock, et al., JAMA 283, 1175 (2000). |
| 35. | R. Baggaley, E. Van Praag, Bull. WHO 78, 1036 (2000). Available online. |
| 36. | F. Dabis, et al., AIDS 14, 1017 (2000). |
| 37. | M. Sinkala, J. P. Stout, S. H. Vermund, R. L. Goldenberg, J. S. Stringer, Lancet 358, 1611 (2001). |
| 38. | M. Bulterys, et al., BMJ 324, 222 (2002). |
| 39. | R. F. Schinazi, A. J. Nahmias, Eds., AIDS in Children, Adolescents & Heterosexual Adults: An Interdisciplinary Approach to Prevention (Elsevier Press, New York, 1988) p. 221. |
| 40. | L. M. Mofenson, Obstet. Gynecol. Clin. North Am. 24, 759 (1997). |
| 41. | M. Lallemant, et al., AIDS 3, 643 (1989). |
| 42. | P. Datta, et al., J. Infect. Dis. 170, 1134 (1994). |
| 43. | R. M. Kumar, S. A. Uduman, A. K. Khurranna, J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 9, 238 (1995). |
| 44. | P. Lepage, et al., Am. J. Epidemiol. 137, 589 (1993). |
| 45. | T. E. Taha, et al., Int. J. Epidemiol. 24, 1022 (1995). |
| 46. | R. J. Biggar, et al., Lancet 347, 1647 (1996). |
| 47. | M. J. Mayaux, et al., J. Pediatr. 131, 857 (1997). |
| 48. | R. J. Simonds, et al., AIDS 12, 301 (1998). |
| 49. | S. Weng, et al., Pediatrics 102, e24 (1998). Available online. |
| 50. | N. Shaffer, et al., Lancet 353, 773 (1999). |
| 51. | R. Nduati, et al., JAMA 283, 1167 (2000). |
| 52. | C. P. Hudson, Int. J. Infect. Dis. 5, 1 (2001). |
| 53. | M. Bulterys, Placenta 22(Suppl. A), S5 (2001). |
| 54. | V. Anderson, et al., Placenta 22(Suppl. A), S34 (2001). |
| 55. | E. Menu, et al., J. Infect. Dis. 179, 44 (1999). |
| 56. | R. E. Dickover, E. M. Garratty, S. Plaeger, Y. J. Bryson, J. Virol. 75, 2194 (2001). |
| 57. | F. O. Aikhionbare, et al., AIDS 15, 2196 (2001). |
| 58. | L. Kuhn, Z. A. Stein, Paediatr. Perinat. Epidemiol. 9, 1 (1995). |
| 59. | G. C. John, J. Kreiss, Epidemiol. Rev. 18, 149 (1996). |
| 60. | M. Bulterys, P. Lepage, Curr. Opin. Pediatr. 10, 143 (1998). |
| 61. | R. L. Edgeworth, K. E. Ugen, Pathobiology 68, 53 (2000). |
| 62. | M. G. Fowler, R. J. Simonds, A. Roongpisuthipong, Pediatr. Clin. North Am. 47, 21 (2000). |
| 63. | The International Perinatal HIV Group, N. Engl. J. Med. 340, 977 (1999). |
| 64. | The European Mode of Delivery Collaboration, Lancet 353, 1035 (1999). |
| 65. | J. P. Ioannidis, et al., J. Infect. Dis. 183, 539 (2001). |
| 66. | The International Perinatal HIV Group, AIDS 15, 357 (2001). |
| 67. | P. Gaillard, et al., AIDS 15, 389 (2001). |
| 68. | P. Msellati, et al., Sex. Transm. Infect. 75, 420 (1999). |
| 69. | M. E. St. Louis, et al., JAMA 269, 2853 (1993). |
| 70. | M. Temmerman, et al., Obstet. Gynecol. 83, 495 (1994). |
| 71. | R. B. Van Dyke, et al., J. Infect. Dis. 179, 319 (1999). |
| 72. | M. Temmerman, et al., Am. J. Obstet. Gynecol. 172, 700 (1995). |
| 73. | D. A. Schwartz, et al., J. Infect. Dis. 182, 1652 (2000). |
| 74. | R. L. Goldenberg, et al., Lancet 352, 1927 (1998). |
| 75. | European Collaborative Study, Lancet 339, 1007 (1992). |
| 76. | R. R. Viscarello, A. B. Copperman, N. J. DeGennaro, Am. J. Obstet. Gynecol. 170, 740 (1994). |
| 77. | S. H. Landesman, et al., N. Engl. J. Med. 334, 1617 (1996). |
| 78. | L. Kuhn, et al., AIDS 11, 429 (1997). |
| 79. | L. Mandelbrot, et al., Am. J. Obstet. Gynecol. 175, 661 (1996). |
| 80. | L. Mandelbrot, et al., JAMA 280, 55 (1998). |
| 81. | P. J. Boyer, et al., JAMA 271, 1925 (1994). |
| 82. | R. S. Sperling, et al., N. Engl. J. Med. 335, 1621 (1996). |
| 83. | R. S. Sperling, et al., AIDS 12, 1805 (1998). |
| 84. | M. Culnane, et al., JAMA 281, 151 (1999). |
| 85. | L. M. Mofenson, et al., N. Engl. J. Med. 341, 385 (1999). |
| 86. | R. Chuachoowong, et al., J. Infect. Dis. 181, 99 (2000). |
| 87. | F. Dabis, et al., Lancet 353, 786 (1999). |
| 88. | S. Z. Wiktor, et al., Lancet 353, 781 (1999). |
| 89. | V. Leroy, et al., AIDS 16, 631 (2002). |
| 90. | M. Lallemant, et al., N. Engl. J. Med. 343, 982 (2000). |
| 91. | J. Saba, paper presented at the 6th Conference on Retrovirus and Opportunistic Infections, Chicago, IL, February 1999. |
| 92. | J. M. Lange for the PETRA trial study team, abstract 17 presented at the 3rd Conference on Global Strategies for the Prevention of HIV Transmission from Mothers to Infants, Kampala, Uganda, September 2001. |
| 93. | L. A. Guay, et al., Lancet 354, 795 (1999). |
| 94. | S. H. Eshleman, et al., AIDS 15, 1951 (2001). |
| 95. | D. Moodley on behalf of the SAINT Investigators Team, abstract LbOr2 presented at the 13th International AIDS Conference, Durban, South Africa, July 2000. |
| 96. | S. Z. Wiktor, et al., abstract TuOrB354 presented at the 13th International AIDS Conference, Durban, South Africa, July 2000. |
| 97. | M. Owor, et al., abstract LbOr1 presented at the 13th International AIDS Conference, Durban, South Africa, July 2000. |
| 98. | K. M. Ayers, D. Clive, W. E. Tucker, G. Hajian, P. De Miranda, Fundm. Appl. Toxicol. 26, 148 (1996). |
| 99. | O. A. Olivero, et al., J. Natl. Cancer Inst. 89, 1602 (1997). |
| 100. | I. C. Hanson, et al., J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 20, 463 (1999). |
| 101. | S. Blanche, C. Rouzioux, L. Mandelbrot, paper presented at the 6th Conference on Retrovirus and Opportunistic Infections, Chicago, IL, February 1999. |
| 102. | S. Blanche, et al., Lancet 354, 1084 (1999). |
| 103. | Perinatal Safety Review Working Group, J. Acquir. Immune Defic. Syndr. 25, 261 (2000). |
| 104. | M. Bulterys, et al., Ann. N. Y. Acad. Sci. 918, 212 (2000). |
| 105. | E. R. Ekpini, et al., Lancet 349, 1054 (1997). |
| 106. | V. Leroy, et al., Lancet 352, 597 (1998). |
| 107. | D. Mbori-Ngacha, et al., JAMA 286, 2413 (2001). |
| 108. | R. Nduati, et al., Lancet 357, 1651 (2001). |
| 109. | A. Coutsoudis, et al., Lancet 354, 471 (1999). |
| 110. | A. Coutsoudis, et al., AIDS 15, 379 (2001). |
| 111. | P. G. Miotti, et al., JAMA 282, 744 (1999). |
| 112. | G. C. John, et al., J. Infect. Dis. 183, 206 (2001). |
| 113. | R. D. Semba, et al., J. Infect. Dis. 180, 93 (1999). |
| 114. | K. Pillay, et al., J. Infect. Dis. 183, 653 (2001). |
| 115. | L. Kuhn, et al., AIDS 15, 1 (2001). |
| 116. | L. Kuhn, S. Meddows-Taylor, G. Gray, C. Tiemessen, Clin. Infect. Dis. 34, 267 (2002). |
| 117. | M. G. Fowler, J. Bertolli, P. Nieburg, JAMA 282, 781 (1999). |
| 118. | H. M. Coovadia, A. Coutsoudis, AIDScience 1, 4 (2001). Available online. |
| 119. | M. Bulterys, et al., AIDS 7, 1639 (1993). |
| 120. | M. Lallemant, et al., AIDS 8, 1451 (1994). |
| 121. | P. B. Matheson, et al., AIDS 10, 1249 (1996). |
| 122. | M. Bulterys, et al., J. Acquir. Immune Defic. Syndr. 15, 76 (1997). |
| 123. | D. N. Burns, et al., J. Infect. Dis. 175, 1206 (1997). |
| 124. | C. M. Wike, et al., AIDS Res. Hum. Retrovir. 8, 1297 (1992). |
| 125. | T. Zhu, et al., J. Virol. 70, 3098 (1996). |
| 126. | L. A. Panther, et al., J. Infect. Dis. 181, 555 (2000). |
| 127. | G. Myers, B. Korber, in The Evolutionary Biology of Viruses, S. S. Morse, Ed. (Raven Press, New York, 1994), pp 211-232. |
| 128. | J. H. Kim, et al., J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 12, 329 (1996). |
| 129. | M. Salminen, AIDS Rev. 2, 178 (2000). |
| 130. | B. Renjifo, et al., J. Hum. Virol. 4, 16 (2001). |
| 131. | W. Fawzi, et al., AIDS 15, 1157 (2001). |
| 132. | M. Bulterys, J. J. Goedert, AIDS 10, 1287 (1996). |
| 133. | "Perinatal HIV Prevention Program," Centers for Disease Control and Prevention [online]. Available online. |
| 134. | C. J. Newschaffer, J. Cocroft, W. W. Hauck, T. Fanning, B. J. Turner, Obstet. Gynecol. 91, 885 (1998). |
| 135. | Reducing the Odds: Preventing Perinatal Transmission of HIV in the United States, M. A. Stoto, D. A. Almario, M. C. McCormick, Eds. (National Academy Press, Washington, 1999). Available online. |
| 136. | N. A. Wade, et al., N. Engl. J. Med. 339, 1409 (1998). |
| 137. | N. A. Wade, G. S. Birkhead, P. T. French, N. Engl. J. Med. 340, 1042 (1999). |
| 138. | G. S. Birkhead, et al., Am. J. Obstet. Gynecol. 183, 245 (2000). |
| 139. | K. K. Van Rompay, et al., AIDS Res. Hum. Retrovir. 14, 761 (1998). |
| 140. | K. K. Van Rompay, et al., J. Virol. 74, 1767 (2000). |
| 141. | K. K. Van Rompay, et al., J. Infect. Dis. 184, 429 (2001). |
| 142. | H. Minkoff, M. J. O'Sullivan, JAMA 279, 1743 (1998). |
| 143. | B. Branson, AIDS Rev. 2, 76 (2000). |
| 144. | M. Bulterys, et al., abstract MoOrC243 presented at the 13th International AIDS Conference, Durban, South Africa, July 2000. |
| 145. | J. S. Stringer, D. J. Rouse, Obstet. Gynecol. 94, 34 (1999). |
| 146. | W. A. Grobman, P. M. Garcia, Am. J. Obstet. Gynecol. 181, 1062 (1999). |
| 147. | M.-L. Newell, et al., AIDS 12, 1571 (1998). |
| 148. | W. J. Graham, M. L. Newell, Lancet 353, 836 (1999). |
| 149. | R. M. Anderson, R. M. May, T. W. Ng, J. T. Rowley, Phil. Trans. R. Soc. Lond. 336, 135 (1992). |
| 150. | M. Bulterys, et al., AIDS 8, 1585 (1994). |
| 151. | M. Laga, Lancet 346, 518 (1995). |
| 152. | M. Laga, B. Schwartlander, E. Pisani, P. S. Sow, M. Carael, AIDS 15, 931 (2001). |
| 153. | S. Kanshana, AIDS 14, 1617 (2000). |
| 154. | A. J. Ammann, S. Nogueira, BMJ 324, 184 (2002). |
| 155. | P. R. Lamptey, BMJ 324, 207 (2002). |
| 156. | T. M. Painter, et al., unpublished data. |
| 157. | T. M. Painter, Soc. Sci. Med. 53, 1397 (2001). |
| 158. | P. Farmer, et al., Lancet 358, 404 (2001). |
| 159. | A. Rosenfield, E. Figdor, Am. J. Public Health 91, 703 (2001). |
| 160. | "The MTCT-Plus Initiative to expand HIV/AIDS care in resource-Limited Settings in Africa, Asia and Latin America," Columbia University Mailman School of Public Health [online]. Available online. |
| 161. | E. Mouzin, E. Mercier, P. Henderson, abstract 332 presented at the 3rd Conference on Global Strategies for the Prevention of HIV Transmission from Mothers to Infants, Kampala, Uganda, September 2001. |
| 162. | D. B. Jelliffe, E. F. Jelliffe, Human Milk in the Modern World (Oxford University Press, Oxford, 1977). |
| 163. | United Nations Special Session on HIV/AIDS [online], New York City, June 2001. Available online. |
| 164. | P. Jha, et al., Science 292, 224 (2001). |
| 165. | L. M. Mofenson, J. A. McIntyre, Lancet 355, 2237 (2000). |
| 166. | J. L. Sullivan, K. Luzuriaga, N. Engl. J. Med. 345, 1568 (2001). |
| 167. | The authors wish to thank Halima Dao, Anniek De Baets, Louise Kuhn, Mary Lou Lindegren, Thomas Painter, R. J. Simonds, and Thomas Welty for their thoughtful comments on this paper. |
| 168. | M. Rogers, unpublished data. |
| Copyright Information | Site map |